Once again recognized by national authorities - Tianjiquan Group is included in the "China Drug Administration Yearbook" (2020)
 2022-04-19
2022-04-19
Recently, Tianjiquan Group has been included in the "China Drug Administration Yearbook" (2020) edited and published by the State Administration for Market Regulation. Previously, Tianjiquan Group had been included in the book multiple times. This not only demonstrates the outstanding achievements of Tianjiquan Group in developing innovative products and the strong appeal of the brand, but also once again proves the authoritative recognition of Tianjiquan Group by the National Medical Products Administration, which is of great significance!

China Drug Administration Yearbook (2020)

China Drug Administration Yearbook (2020)
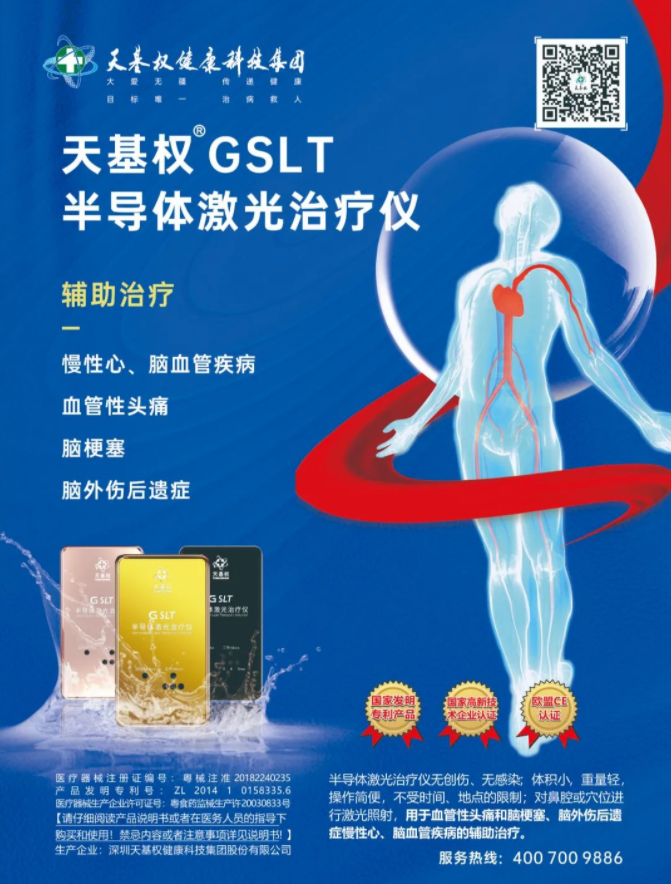
The book "China Drug Administration Yearbook" (2020) focuses on the Tianjiquan G SLT semiconductor laser therapy device. The Tianjiquan G SLT semiconductor laser therapy device uses the principle of low-energy laser irradiation to emit low-intensity 650 ± 10nm weak laser. Through nasal irradiation, it improves cell metabolism, corrects abnormal cell metabolism, accelerates the clearance of oxygen free radicals, thereby improving blood microcirculation, regulating cell immunity, protecting target organ function, and reducing the probability of various complications. It is used as an adjuvant therapy for various cardiovascular and cerebrovascular diseases. The G SLT semiconductor laser therapy device is a physical therapy for extracorporeal blood purification, without the various toxic side effects caused by drugs passing through organs. It is a very mature, safe, and effective device for treating cardiovascular and cerebrovascular diseases and preventive healthcare. It has been certified by the National Food and Drug Administration and the European Union CE certification.
China Drug Administration Yearbook (2020) Tianjiquan Page Details
Tianjiquan has an independent research and development production base to provide guarantees for technology research and product testing. We adhere to using innovative technology to create ultimate health products, and have developed multiple health technology products that have been officially launched on the Shenzhen government procurement platform. In addition, with its high-precision technology, Tianjiquan Group has also won multiple honorary titles such as "EU CE Certification", "Medical Device Import License" from South Korea, "National High tech Enterprise", "China's Most Influential Innovation Achievement Award", "South Africa Outstanding Science and Technology Achievement Award", "Thailand Inventor's Day China Good Technology Exhibition Gold Award", and has won the Excellent Product Award at multiple high-tech fairs in 2010, 2011, 2012, etc.
Since its establishment more than 20 years ago, Tianjiquan Group has always adhered to the principle of "boundless love, spreading health; unique goal, treating diseases and saving lives." With a down-to-earth approach, it has adapted to the market and innovated, accumulating rich experience and strong strength. The inclusion of the book "China Drug Administration Yearbook" (2020) is a great event for the development of Tianjiquan Group's business, a blessing for the inheritance of corporate culture, and another milestone representing Tianjiquan Group's efforts to help the big health industry reach a new level.
In the future, Tianjiquan Group will live up to the high expectations of the country and the people, always adhere to its original mission, strive to improve its scientific and technological research and development level, develop more and better health products to serve the public, fully leverage the advantages and exemplary role of high-tech enterprises, never forget its original aspiration, forge ahead, and truly realize the Tianjiquan dream of making the people of the whole country healthy!